Medical Device
Consultancy Service -
Enable Your Regulatory Compliance Through Our Knowledge, Skills & Systems
Your consultancy and QMS partner in the medical device industry
As medical device experts we understand both your regulatory and commercial needs
We are a team of experienced regulatory and compliance experts specialising in medical devices. We partner with medical device companies to enhance their quality management systems and obtain regulatory compliance that fits the needs of their medical device and the international regions they are targeting.
We are passionate about the medical device industry. We see that its progression and its advances in technology benefit society. At MedQtech we are keen to help our customers in that journey of progression, accelerating the process wherever possible.
Our Values
True customer partnership
The human touch – Because we care
Delivering business value
Consultancy & systems together
Enabling innovation effectively
Dedication to improvement
If you are asking these questions we can help you in driving regulatory compliance and quality management in your business

What classification rules apply for our medical device?
How does our quality system get ISO13485 certified?
How do I start my regulatory compliance journey?
Are we ready for the new MDR/IVDR requirements?
What technical documentation do I have to have in place?
Who can support us with the regulatory compliance process?
Medical device specialist consultancy – learn from our industry knowledge, gain momentum from our industry passion.
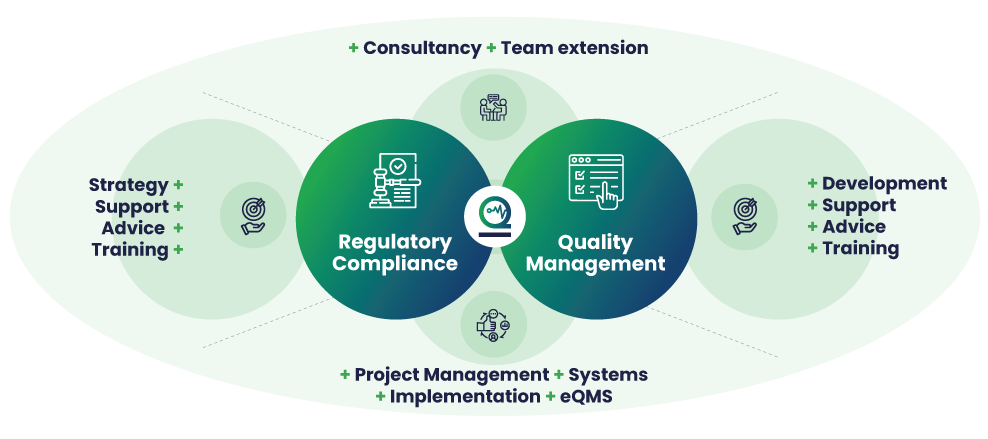
As a team we can offer you the correct consultancy services that can enable your business with regulatory compliance and quality management. Importantly we offer consultancy services such as strategy advice, training and project management and we can also assist you with implementing your Quality Management System – learn more about our eQMS solution here >
“We partnered with MedQtech because their experience and knowledge complimented ours, together we have achieved our goals more effectively and raised our quality systems to benefit the whole organisation. We have an ongoing relationship with MedQtech as an advisory partner”
Medical device company